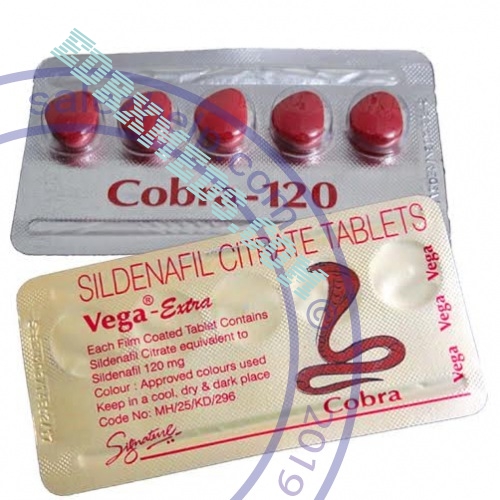
The pharmacokinetics of Vegal Extra in the recommended dose range is linear.
Suction. After intake of Vegah Cobra is rapidly absorbed. Absolute bioavailability averages about 40% (25 to 63%). In vitro Vegal Extra at a concentration of about 1.7 ng/ml (3.5 nm) inhibits the activity of PDE-5 50%. After a single dose of Vegal Extra 100 mg average Cmax free Vegal Extra in plasma men is about 18 ng/ml (38 nm). Cmax when taking Vegal Extra inside fasting is achieved for an average of 60 minutes (from 30 to 120 minutes). When taken in combination with fatty foods, the rate of absorption decreases: Cmax decreases by an average of 29%, and Tmax increases by 60 minutes, but the degree of absorption does not change significantly (AUC decreases by 11%).
Distribution. The average Vss of Vega Cobra 120 Mg is 105 l. the Association of Cobra Vega 120 and its main circulating N-demethyl metabolite with plasma proteins is about 96% and does not depend on the total concentration of the drug. Less than 0.0002% of the dose of Vegal Extra (an average of 188 ng) was found in sperm 90 minutes after taking the drug.
Metabolism. Vegal Extra is metabolized mainly in the liver by the action of CYP3A4 isoenzyme (main pathway) and CYP2C9 isoenzyme (minor pathway). The main circulating active metabolite, formed as a result of N-demethylation of Cobra Vega, undergoes further metabolism. The selectivity of this metabolite against PDE is comparable to that of Cobra Vega 120 Mg, and its activity against PDE-5 in vitro is about 50% of the activity of Vegal Extra. The concentration of the metabolite in the blood plasma of healthy volunteers was about 40% of the concentration of Sildenafil Tablets Vegah Extra Cobra. N-demethyl metabolite undergoes further metabolism; its T1/2 is about 4 no.
Breeding. The total clearance of Signature Cobra Vega is 41 l/h, and the final T1/2 — 3-5 h. After oral administration, as well as after I/V, Vegah Extra 130 is excreted as metabolites, mainly by the intestine (about 80% oral dose) and to a lesser extent by the kidneys (about 13% oral dose).
Dosage and administration
Inside, can be taken with or without water. When taking the drug, you should put a tablet, dispersed in the oral cavity, on the tongue, after which it will quickly dissolve, and it can be swallowed. The tablet should be taken immediately after opening the blister. Patients who are recommended a dose of Vega Visa 100 mg, the second tablet of Sildenafil Tablets Vega Extra Cobra 50 mg should be taken after complete dissolution of the first tablet.
The recommended dose for most adult patients is 50 mg approximately 1 hour before sexual activity. Taking into account the effectiveness and tolerability of the dose can be increased to 100 mg or reduced to 25 mg (only tablets coated with a film, the appropriate dosage). The maximum recommended dose is 100 mg. Patients who are recommended a dose of Vegal Extra 100 mg, it is necessary to take 2 table., dispersible in the oral cavity, dosage 50 mg consecutively one after another. The maximum recommended frequency of use — 1 time per day. It should be borne in mind that the absorption of Vegal Extra significantly slows down when used in combination with fatty foods.
Special patient groups
Renal impairment. In mild to moderate renal failure dose adjustment is not required, in severe renal failure (Creatinine CL <30 ml/min) Sildenafil Vega Extra 130 dose should be reduced to 25 mg.
Hepatic impairment. Since the excretion of Cobra Vega Extra Strong 120 Mg is disturbed in patients with liver damage (in particular, cirrhosis), the dose of Vega Cobra 120 should be reduced to 25 mg.
Joint application with other drugs. Joint use with ritonavir is not recommended. In any case, the maximum dose of Vegal Extra under any circumstances should not exceed 25 mg, and the frequency of application — 1 time in 48 h (see "Interaction").
When combined with inhibitors of CYP3A4 isoenzyme (erythromycin, saquinavir, ketoconazole, Itraconazole), the initial dose of Vegal Extra should be 25 mg (see "Interaction").
To minimize the risk of postural hypotension in patients taking α-blockers, Vegal Extra should be started only after achieving hemodynamic stabilization in these patients. Consideration should also be given to reducing the initial dose of Vegah Extra 120 Mg (see special instructions and Interactions).
Old age. Adjustment of the dose of Vegal Extra is not required.
Hearing impairment
Some post-marketing and clinical studies have reported cases of sudden hearing loss or impairment associated with the use of all PDE-5 inhibitors, including Vegal Extra. Most of these patients had risk factors for sudden impairment or hearing loss. There is no causal relationship between the use of PDE-5 inhibitors and sudden hearing impairment or hearing loss. In case of sudden hearing impairment or hearing loss while taking Vegal Extra, consult a doctor immediately.
Bleedings
Vega Extra 120 enhances the antiplatelet effect of sodium nitroprusside, a nitric oxide donator, on human platelets in vitro. Data on the safety of Vega Coin in patients with a tendency to bleeding or exacerbation of gastric ulcer and duodenal ulcer are not available, so the drug Vegal Extra in these patients should be used with caution (see With caution).
The incidence of nosebleeds in patients with pulmonary hypertension associated with diffuse connective tissue diseases was higher (Vegal Extra — 12.9%, placebo — 0%) than in patients with primary pulmonary arterial hypertension (Vega Visa — 3%, placebo — 2.4%). Patients receiving Vegal Extra in combination with an antagonist of vitamin K, the frequency of nasal bleeding was higher (8.8 percent) than patients who were not taking an antagonist of vitamin K (1.7 percent).
Use in conjunction with other means for the treatment of erectile dysfunction
The safety and efficacy of Vegal Extra together with other PDE-5 inhibitors or other drugs for the treatment of pulmonary hypertension containing Vegal Extra (eg, Revatsio®), or other means for the treatment of erection disorders have not been studied, so the use of such combinations is not recommended (see "Contraindications").
Influence on the ability to drive and mechanisms. In patients receiving Signature Cobra Vega any negative effect on the ability to drive a car or other means was not observed. However, since when taking Vegal Extra may develop dizziness, lowering blood PRESSURE, the development of chromatopsia, blurred vision, etc. side effects, care should be taken when driving and doing other potentially dangerous activities that require increased concentration and speed of psychomotor reactions. Also, you should be careful about the individual action of the drug in these situations, especially at the beginning of treatment and when changing the dosage regimen.
Cardiovascular complications
During the post-marketing use of Extra Vega for the treatment of erectile dysfunction, adverse events such as severe cardiovascular complications (including myocardial infarction, unstable angina, sudden cardiac death, ventricular arrhythmia, hemorrhagic stroke, transient ischemic attack, hypertension and hypotension) were reported, which had a temporary connection with the use of Vegal Extra. Most of these patients, but not all of them, had risk factors for cardiovascular complications. Many of these adverse events were observed shortly after sexual activity, and some were observed after taking Vegab Extra without subsequent sexual activity. It is not possible to establish a direct link between the reported adverse events and those or other factors.
Hypotension
Vegal Extra has a systemic vasodilating effect, leading to a transient decrease in blood PRESSURE, which is not clinically significant and does not lead to any consequences in most patients. However, before prescribing Vegab Extra, the doctor should carefully assess the risk of possible undesirable manifestations of vasodilating action in patients with relevant diseases, especially against the background of sexual activity. Increased susceptibility to vasodilators is observed in patients with obstruction of the output tract of the left ventricle (aortic stenosis, GOCMP), as well as with the rare syndrome of multiple systemic atrophy, manifested by a severe violation of the regulation of blood PRESSURE from the autonomic nervous system.
Since the combined use of Signature Cobra Vega and α-blockers can lead to symptomatic hypotension in some sensitive patients, Vega Cobra should be used with caution in patients taking α-blockers (see "Interaction"). To minimize the risk of postural hypotension in patients taking α-blockers, the drug Signature Cobra Vega should be started only after achieving stabilization of hemodynamic parameters in these patients. It should also consider reducing the initial dose of Vega Extra 120 Mg (see. "Dosage and administration"). The doctor should inform patients about what actions should be taken in case of symptoms of postural hypotension.
Vega Pills