Clinical data
In a placebo-controlled cross-examination of patients with proven early age macular degeneration (n=9), Sildenafil Vega Extra 130 was well tolerated at a single dose of 100 mg. There were no clinically significant changes in vision evaluated by special visual tests (visual acuity, Amsler lattice, color perception, color simulation, Humphrey perimeter and photostress).
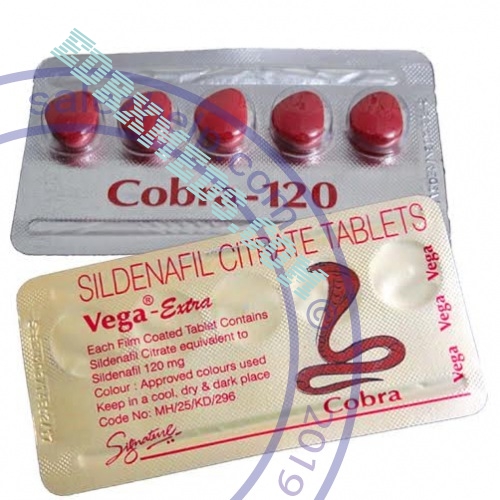
Efficiency. The efficacy and safety of Cobra Vega 120 Mg were evaluated in 21 randomized double-blind placebo-controlled studies lasting up to 6 months in 3000 patients from 19 to 87 years with erectile dysfunction of different etiology (organic, psychogenic or mixed). The efficacy of the drug was evaluated globally using an erection diary, an international erectile function index (validated questionnaire on the state of sexual function) and a partner survey.
The effectiveness of Sildenafil Vega Extra 130, defined as the ability to achieve and maintain an erection sufficient for satisfactory sexual intercourse, has been demonstrated in all studies and confirmed in long-term studies lasting 1 year. In studies with the use of fixed-dose proportion of patients reporting that treatment improved their erections were 62% (dose of Sildenafil Vega Extra 130 25 mg), 74% (dose Vega Pills 50 mg) and 82% (dose Sildenafil Vega Extra 130 100 mg) compared to 25% in the placebo group. Analysis of the international index of erectile function showed that in addition to improving erection treatment Sildenafil Vega Extra 130 also increased the quality of orgasm, allowing to achieve satisfaction from sexual intercourse and overall satisfaction.
According to the generalized data, 59% of patients with diabetes, 43% of patients who underwent radical prostatectomy and 83% of patients with spinal cord injuries (against 16, 15 and 12% in the placebo group, respectively) were reported to have improved erection in the treatment of Sildenafil Vega Extra 130.
Pharmacokinetics
Vega Extra is a potent selective inhibitor of cGMP-specific PDE-5.
Mechanism of action
The implementation of the physiological mechanism of erection is associated with the release of nitric oxide (NO) in the cavernous body during sexual stimulation. This, in turn, leads to an increase in the level of cGMP, the subsequent relaxation of smooth muscle tissue of the cavernous body and an increase in blood flow.
Sildenafil Vega Extra 130 does not have a direct relaxing effect on the isolated cavernous human body, but enhances the effect of nitric oxide by inhibiting PDE-5, which is responsible for the disintegration of cGMP.
Selective Vega Extra Strong 120 against PDE5 in vitro, its activity against PDE5 superior activity against other known PDE isoenzymes: PDE-6 — 10 times; PDE-1 more than 80 times; PDE-2, PDE-4, PDE-7 PDE–11 more than 700 times. Vega Extra 120 Mg is 4000 times more selective for PDE-5 than PDE-3, which is crucial because PDE-3 is one of the key enzymes in the regulation of myocardial contractility.
A prerequisite for the effectiveness of Sildenafil Vega Extra 130 is sexual stimulation.
Sildenafil Vega Extra 130 restores impaired erectile function in conditions of sexual stimulation by increasing blood flow to the cavernous bodies of the penis.
Dosage and administration
Inside, can be taken with or without water. When taking the drug, you should put a tablet, dispersed in the oral cavity, on the tongue, after which it will quickly dissolve, and it can be swallowed. The tablet should be taken immediately after opening the blister. Patients who are recommended a dose of Sildenafil Vega Extra 130 100 mg, the second tablet of Extra Vega 50 mg should be taken after complete dissolution of the first tablet.
The recommended dose for most adult patients is 50 mg approximately 1 hour before sexual activity. Taking into account the effectiveness and tolerability of the dose can be increased to 100 mg or reduced to 25 mg (only tablets coated with a film, the appropriate dosage). The maximum recommended dose is 100 mg. Patients who are recommended a dose of Sildenafil Vega Extra 130 100 mg, it is necessary to take 2 table., dispersible in the oral cavity, dosage 50 mg consecutively one after another. The maximum recommended frequency of use — 1 time per day. It should be borne in mind that the absorption of Vegab Extra significantly slows down when used in combination with fatty foods.
Special patient groups
Renal impairment. In mild to moderate renal failure dose adjustment is not required, in severe renal failure (Creatinine CL <30 ml/min) Sildenafil Vega Extra 130 dose should be reduced to 25 mg.
Hepatic impairment. Since the excretion of Vegah Extra 120 Mg is disturbed in patients with liver damage (in particular, cirrhosis), the dose of Sildenafil Vega Extra 130 should be reduced to 25 mg.
Joint application with other drugs. Joint use with ritonavir is not recommended. In any case, the maximum dose of Sildenafil Vega Extra 130 under any circumstances should not exceed 25 mg, and the frequency of application — 1 time in 48 h (see "Interaction").
When combined with inhibitors of CYP3A4 isoenzyme (erythromycin, saquinavir, ketoconazole, Itraconazole), the initial dose of Sildenafil Vega Extra 130 should be 25 mg (see "Interaction").
To minimize the risk of postural hypotension in patients taking α-blockers, Sildenafil Vega Extra 130 should be started only after achieving hemodynamic stabilization in these patients. Consideration should also be given to reducing the initial dose of Sildenafil Vega Extra 130 Mg (see special instructions and Interactions).
Old age. Adjustment of the dose of Vegah Extra 120 Indication is not required.
Visual impairment
In rare cases, during post-approval use of all of the inhibitors of PDE5, including Sildenafil Vega Extra 120, reported NESN is a rare disease and the cause of the decrease or loss of vision. Most of these patients had risk factors, such as decreased ratios of excavation diameter to optic disc (stagnant disc), age over 50, diabetes, hypertension, CHD, hyperlipidemia, and Smoking. In an observational study evaluated whether recent use of drugs class of inhibitors PDE-5 with an acute onset NESN. The results indicate approximately a 2-fold increase in risk NESN within 5T1/2 after application of the PDE-5 inhibitor. According to published literature, the annual incidence of npins IS 2.5–11.8 cases per 100,000 men aged ≥50 years in the General population. Patients should be advised to stop Sildenafil Vega Extra 130 therapy in case of sudden loss of vision and consult a doctor immediately.
Persons who have already had a case NESN have an increased risk of relapse NESN. Therefore, the doctor should discuss this risk with such patients, as well as the potential chance of adverse effects of PDE-5 inhibitors. PDE-5 inhibitors, including Sildenafil Vega Extra 130, in such patients should be used with caution and only in situations where the expected benefit outweighs the risk. In patients with episodes of development PINZN with loss of vision in one eye receiving Sildenafil Vega Extra 130 is contraindicated (see "Contraindications").
A small number of patients with hereditary retinitis pigmentosa have genetically determined disorders of the PDE of the retina. Information about the safety of the drug Sildenafil Vega Extra 130 in patients with retinitis pigmentosa absent, so these patients should not be used Sildenafil Vega Extra 130 (see "Contraindications").
Cobra Vega